|
UBC MRI CENTRE SCANNERS
The UBC MRI Research Centre was established to support the local biomedical research community with the resources and expertise necessary to implement MRI techniques in their research. The main centrepiece of our preclinical research capabilities is a 7 TESLA MRI SCANNER designed specifically for small experimental animals such as mice and rats. We take pride in providing a full range of research support for MR imaging and spectroscopy applications including method development, data collection and data analysis. Our approach is very much a collaborative one, with user research driving new technique development and vice versa. Interested researchers and industry clients are encouraged to submit MR proposals to the Centre; our staff will be happy to provide advice and help to plan your scanning protocols. Highlights of our facility infrastructure include:
 |
- Bruker Biospec 7 Tesla MRI scanner (4 receiver channels, 20 cm-diameter accessible bore size)
- In-scan isofluorane anesthesia system and body temperature control
- Monitoring system for vital signs (temperature, respiration and ECG)
- Animal prep room for scan preparation and small surgeries
- Electronics/machining workshop and computing facilities
- Three full-time staff including a physicist, engineer and scanner operator
|
MORPHOLOGICAL IMAGING: HIGH RESOLUTION AT ARBITRARY ORIENTATIONS
FUNCTIONAL IMAGING: INVESTIGATING BIOLOGICAL PROCESSES
Apart from morphological imaging, current MRI applications have tended towards measuring the function of biological systems, which further clarifies the "mechanism of action" which underlie a disease process or therapeutic intervention. For example, magnetic resonance spectroscopy (MRS) can be used to study metabolic processes through measurement of in vivo metabolite concentrations: it is possible to detect high-energy phosphates and hydrogen-based metabolites in brain, heart, muscle and tumour.
 |
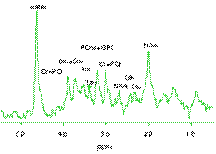 |
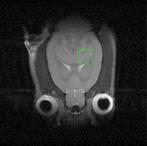
| In vivo rat brain image (left) and proton metabolite spectrum acquired in hippocampus (right) |
Vascular parameters such as blood flow and vessel permeability can also be investigated, most commonly through rapid monitoring of signal enhancement while an intravenously injected contrast agent passes through the organ of interest. Finally, one technique that has gained great popularity is the so-called "functional MRI" (fMRI) technique, where MRI signal changes in the nervous system reflect increases in local blood flow, which are interpreted as the effect of local neurological activity. fMRI has gained wide acceptance in the human neuroscience and psychology fields; however its application to animal models is still a matter of ongoing development.
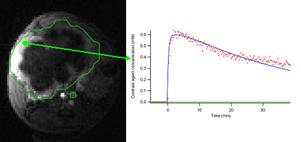
Dynamic contrast-enhanced MRI of orthotopic mouse model of pancreatic cancer.
Inset shows time course of contrast agent concentration, which can be fit to models reflecting blood flow and vessel permeability. |
MOLECULAR IMAGING: DETECTION OF CELLULAR EVENTS AND BEYOND
The current frontier in preclinical MRI research is "molecular imaging", which aims to noninvasively detect events on the cellular and subcellular level. This approach typically involves attaching a payload of MRI-visible contrast agent to a cell or nanoparticle that would otherwise be undetectable under MRI. In this way, therapeutic agents such as drugs, lentiviral vectors, and stem cells can be "labeled" in order to monitor the biodistribution and pharmacokinetics of the agent following injection. Another approach is to conjugate the MRI contrast agent with a ligand which preferentially binds to specific endogenous receptors, opening up the possibility to detect the expression of enzymes or receptors that arise in specific biochemical pathways. By far, the biggest challenge with using MRI for molecular imaging is its low sensitivity; MRI contrast agent concentrations must at least be in the micromolar range, whereas radiotracer contrast agents used in positron emission tomography (PET) can be detected in the picomolar range. However, MRI does provide the advantage of acquiring images that elucidate both the "molecular" events as well as the anatomy in the same experiment with the same instrument.
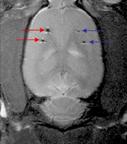
Image of rat brain implanted with retinal epithelial cells: labeled (red arrows) and unlabeled (blue arrows) |
Please contact Imaging
(imagingmri@bcprc.ca) for more information.
|













